About
The Data Space Platform for real-world data
Get access to real-world clinical data for secondary use, faster and fully compliant.
request your demo
NLP
OMOP CDM
Data Delivery
Data Catalog
Federated Network
this is what we do
IOMED is a Data Space Platform that, thanks to AI and NLP, enables Hospitals and Life Science companies secure and compliant access to real-world clinical data for research purposes.
Added Value
[ 01 ]
AI-powered Clinical Data Activation
We leverage advanced AI and NLP to extract and structure data across all hospital text sources, transforming clinical information into research-ready real-world data. Read more...
[ 02 ]
Full Hospital Deployment, TA- Agnostic by Design
Our full in-hospital deployment ensures consistent data retrieval across sites and systems, enabling research beyond a single disease or predefined use case. Read more...
[ 03 ]
Built-in Validation and Verification
We work with high-quality verification and validation processes to guarantee high data quality and accuracy, cross-site consistency, and compliance-ready real-world data. Read more...
[ 04 ]
EU5 Hospital Partner Network
Go beyond single-center limitations with access to a trusted EU5 hospital network, harmonized under the OMOP Common Data Model to ensure interoperability, representativity, consistency, and reliability. Read more...
[ 05 ]
Faster Feasibility and Evidence Generation
Gain visibility into available data before launching studies or requests, reducing uncertainty and enabling faster, evidence-based feasibility decisions. Read more...
[ 06 ]
Research-Ready Data, On Demand
Submit research-specific data requests through the platform and receive harmonized outputs at patient-level, aggregated insights, or recruitment-ready data. Read more...
[ 07 ]
Manage Hospital Approvals
Initiate study approval requests directly through the platform and manage hospital participation end-to-end. Built-in workflows support ethical approvals, contracts, and formal authorization processes. Read more...
HOW IT WORKS
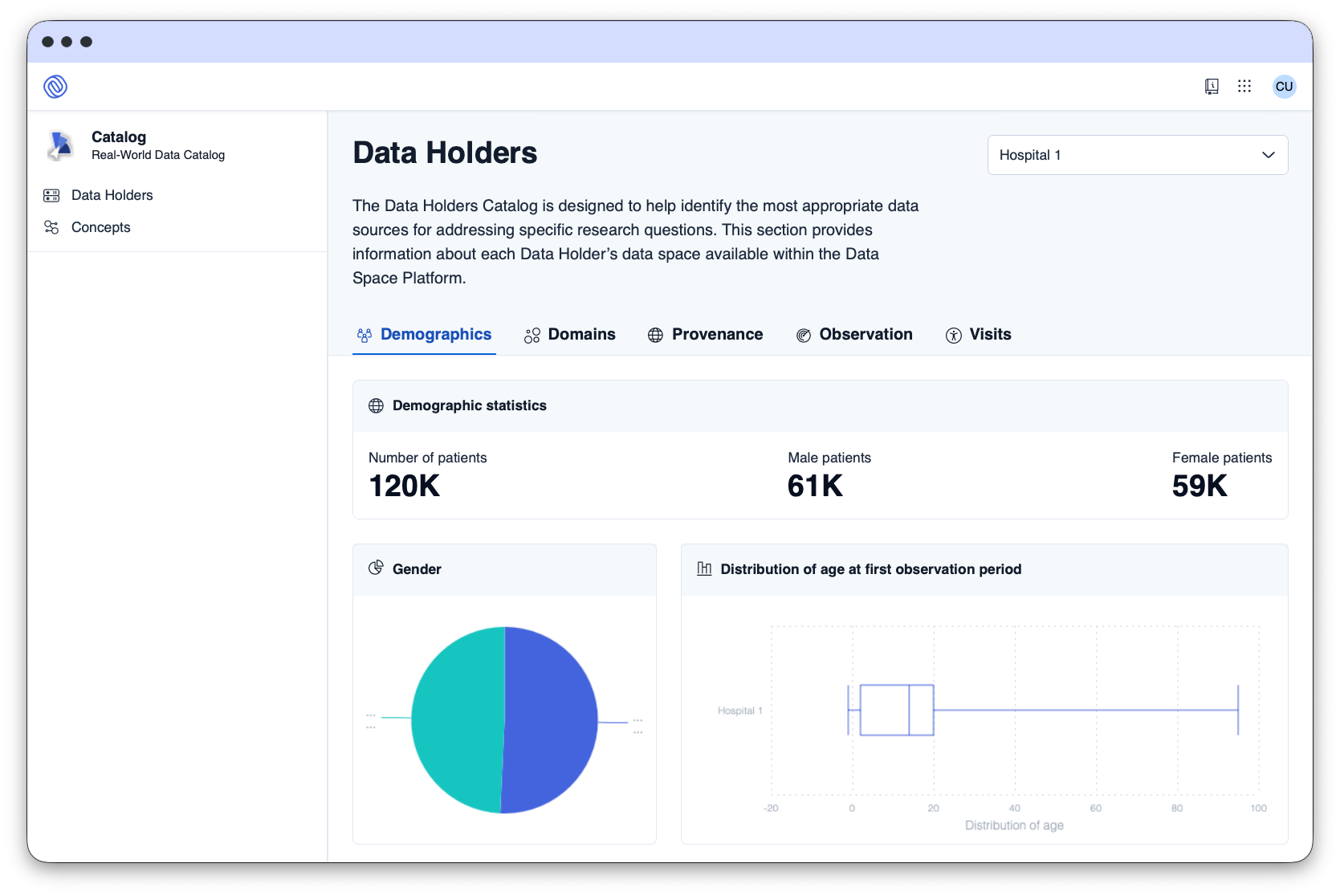
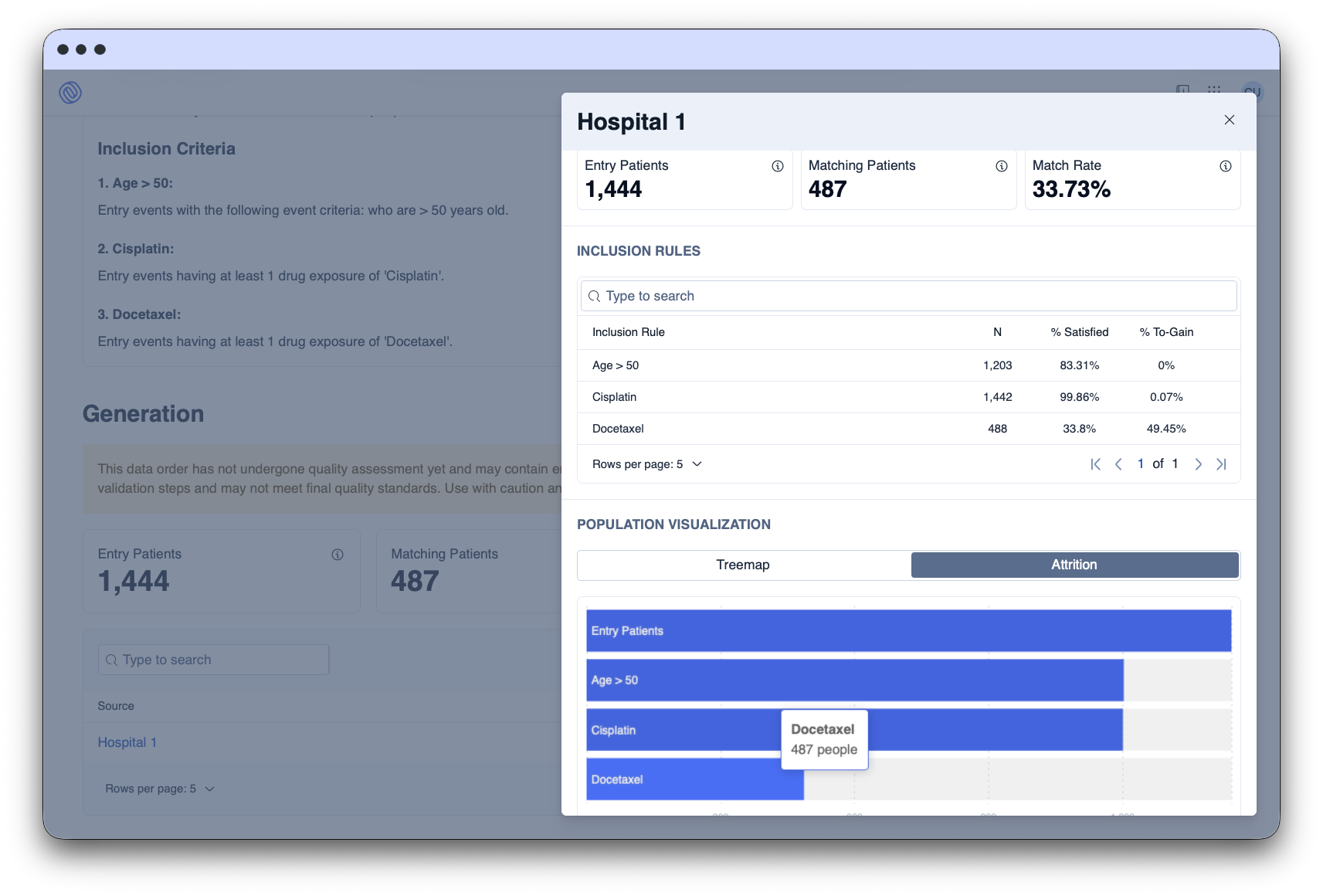
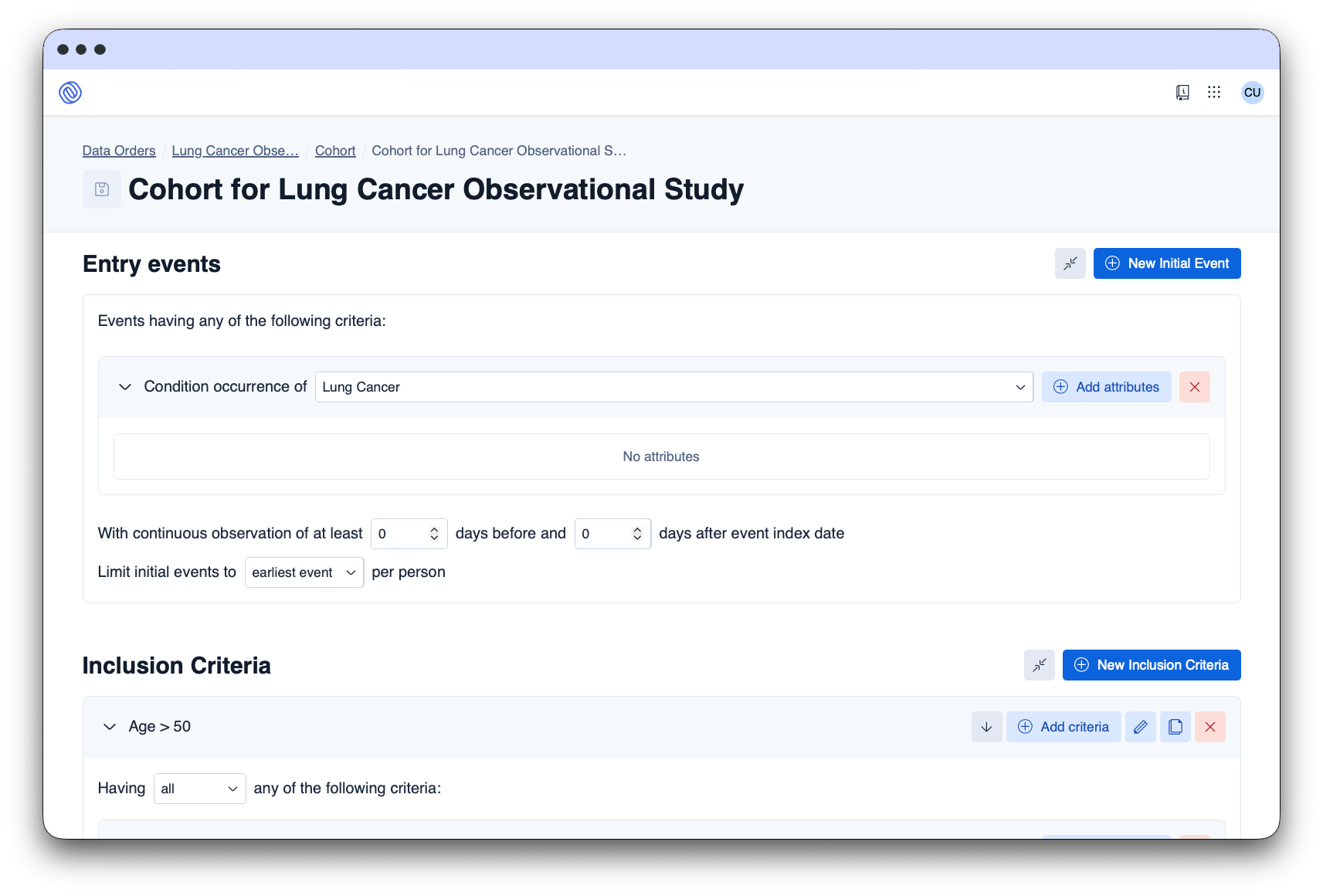
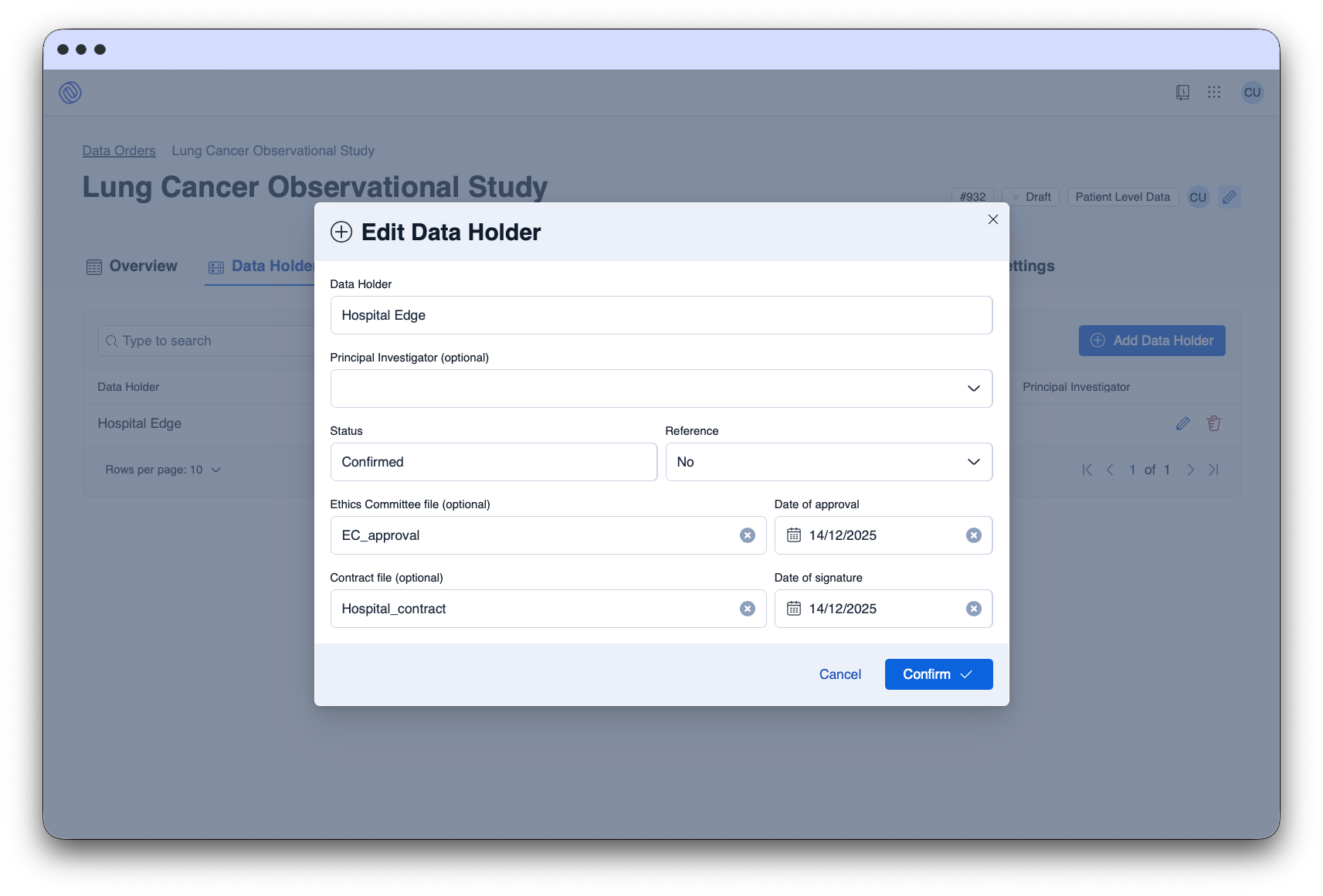
The Data Space Platform facilitates the request, approval, and delivery of data for secondary use through automated processes, ensuring compliance.
BUILT FOR
Hospitals and Healthcare Providers
Get access to your center data ready for research use. Operationalize the secondary use of clinical data with full ownership, governance, and EHDS readiness. Enable your organization to attract multicentric and international research studies.
Life Sciences Companies
Access longitudinal, real-world hospital data on demand, secure, compliant, and research-ready. Generate robust evidence across the full product lifecycle, accelerate study timelines, reduce operational complexity, and activate both structured and unstructured clinical data for high-quality research and evidence generation.
ISO 27701:2019 certified
Privacy-by-design infrastructure for secure and compliant clinical data access

RECOGNITIONS AND CERTIFICATIONS





ISO 9001:2015

ISO 27001:2022

ISO 27701:2019

Top 50 CB Insights

EU Seal of Excellence
.png)
EHDEN Certified SME

OHDSI Partners
What´s New?
No items found.
What´s New?








